Basics of Herpes Viruses for the Ophthalmologists
There are 219 virus species are known to infect humans. The first human virus identified was yellow fever virus in 1901, following earlier discoveries of tobacco mosaic virus in 1892 and foot-and-mouth disease virus in 1898. Yellow fever virus was the first human “filterable agent” to be recognized. Even today, new human viruses continue to be identified at a rate of approximately three to four species per year. In fact, viruses account for more than two-thirds of all newly emerging human pathogens.
Kinetics of Viral Infections
Basic steps of viral infections have been sumarised below with an example of Herpes simplex virus within brackets
Virus attaches to specific receptors on the host cell surface (HSV binds to heparan sulfate and nectin receptors on epithelial cells)→Virus enters the cell, by fusion with the membrane or by endocytosis (HSV envelope fuses with the host cell membrane)→Viral coat (capsid) breaks open and releases its genetic material (HSV capsid travels to the nucleus and releases double-stranded DNA)→Viral genetic material multiplies inside the cell using viral enzymes (HSV DNA replicates in the nucleus using viral DNA polymerase)→New virus particles are assembled (HSV nucleocapsids assemble in the nucleus and acquire envelope)→New viruses leave the cell, often damaging or destroying it (HSV exits by budding and can cause cell lysis, leading to vesicle formation)
- Lytic Infection = An active replication of the viral genome results in a lytic infection characterized by the release of new progeny virus particles, often upon the lysis of the host cell.
- Latent Infection = The virus is 'quiescent' - a state in which the virus is not replicating .
For ophthalmologists, herpes viruses are among the most clinically relevant pathogens, particularly Herpes simplex virus (HSV), Varicella–zoster virus (VZV), and Cytomegalovirus (CMV). Beyond causing a large proportion of ocular infections, what truly distinguishes herpesviruses is their remarkable biological behavior. As James Lovelock aptly stated, “An inefficient virus kills its host. A clever virus stays with it.” Herpesviruses exemplify this principle. Their defining feature is the ability to establish lifelong latency in the natural host. After primary infection, the virus enters specific cells—most notably neurons—and persists in a dormant state. During latency, the viral genome remains within the cell nucleus in a circular episomal form, and only a limited set of viral genes is expressed. The virus does not actively replicate but retains the capacity to reactivate under suitable conditions. Thus, herpesviruses possess two key biological properties: 1. The ability to invade and replicate within the host nervous system. 2. The ability to establish latent infection with lifelong persistence and potential for reactivation. It is this combination of neurotropism and latency that makes herpesviruses uniquely important in ophthalmic practice.
There are over 100 known herpesviruses across species, but only 8-9 routinely infect humans as human herpesviruses (HHV-1 through HHV-8). HHV-1 (HSV-1), HHV-2 (HSV-2), HHV-3 (VZV), HHV-4 (EBV), HHV-5 (CMV), HHV-6, HHV-7, HHV-8 (KSHV). Herpesviridae divides into Alphaherpesvirinae (α), Betaherpesvirinae (β), Gammaherpesvirinae (γ) subfamilies based on replication cycle, host range, and latency sites.
| Alphaherpesviruses | Betaherpesviruses | Gammaherpesviruses |
| HSV1,2 + VZV | CMV | EBV |
HSV, VZV, and CMV—all alpha- and betaherpesviruses—replicate in human cell nuclei but differ fundamentally in cycle speed, cell effects, and gene timing. HSV completes cycles rapidly (~8 hours), VZV (is moderately paced (12-24 hours, skin-focused), while CMV is slowest (3-5 days) (2)
How the names of these three viruses originated :
- HSV : “Herpes” from Greek for “creeping” (vesicular skin rash); “simplex” distinguishes primary oral/genital types (1/2) from varicella.
- VZV (Varicella-Zoster Virus): “Varicella” for chickenpox rash; “zoster” for shingles (Greek “girdle,” belt-like eruption).
- CMV (Cytomegalovirus): Named for large (mega) intranuclear inclusions in enlarged (cyto-) cells, seen in histology.
Brief Summary of HSV infections of Eye
Herpes simplex virus (HSV) is a double-stranded DNA virus belonging to the Alphaherpesvirinae subfamily of the Herpesviridae family, featuring one of the larger viral genomes among human herpesviruses at approximately 152 kbp encoding about 80 genes. Humans serve as the only natural reservoir for HSV types 1 and 2. HSV is an enveloped, heat-labile virus that spreads primarily cell-to-cell via direct contact with infected secretions or lesions, or through short-range airborne droplets.
Primary HSV infection generally occurs early in life, once maternally derived antibodies have waned and no longer provide adequate protection. The initial infection takes place in epithelial cells, where the virus undergoes a lytic cycle. Newly formed viral particles subsequently enter adjacent sensory nerve terminals. The nucleocapsid is then transported along the axon to the neuronal cell body through microtubule-dependent retrograde transport. During replication within the corneal epithelium, HSV-1 gains entry into the sensory nerve endings and travels by retrograde axonal transport to the neuronal nuclei located in the ophthalmic division of the trigeminal ganglion. (3),(4), (5)
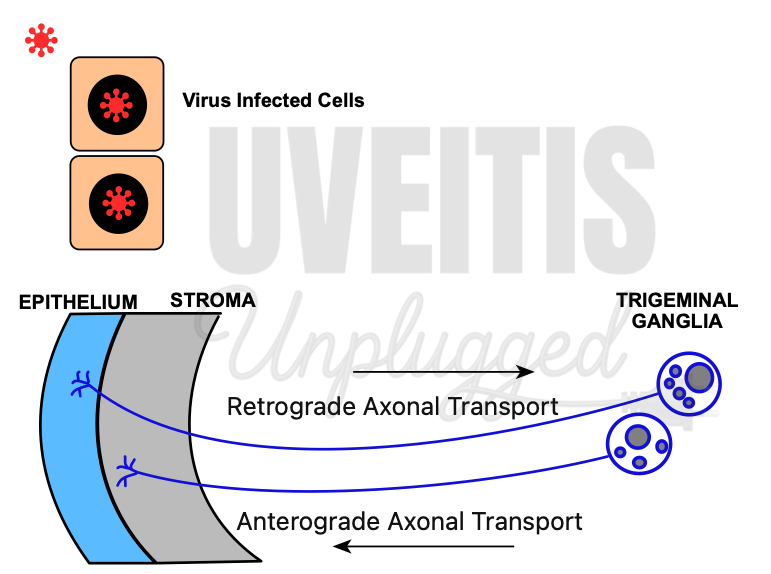
In the latent phase, the viral genome persists within the neuron, but no infectious virions are produced. One or more copies of the HSV-1 genome are maintained in sensory neurons for the lifetime of the host. Replication occurs only in a small proportion of latently infected neurons, and these affected neurons may ultimately undergo cell death. Although circulating antibodies do not prevent reactivation, they help restrict viral spread to adjacent tissues.
Upon reactivation, the viral genome resumes replication. Newly formed capsids are transported by anterograde axonal transport toward the peripheral tissues. In certain individuals, HSV-1 periodically reactivates in a limited number of infected neurons, and viral components travel down the axons to the cornea, where assembly and release occur. (3), (4), (5)
The virus then re-infects epithelial cells, initiating another lytic cycle. Reactivation can be triggered by various stimuli, leading to renewed viral replication and clinical disease. Recurrent viral shedding in the cornea results in repeated episodes of herpes simplex keratitis (HSK), which may progressively cause corneal scarring and visual impairment.
Brief Summary of VZV infections of the Eye:
Varicella–zoster virus (VZV) is a double-stranded DNA virus belonging to the herpesvirus family and possesses one of the smallest genomes among human herpesviruses, measuring approximately 150–200 nm in diameter. Humans are the only known natural reservoir. VZV is heat-labile and spreads predominantly through direct cell-to-cell contact.
Primary VZV infection manifests clinically as chickenpox. Following entry through the respiratory tract, the virus replicates in the respiratory epithelium and regional lymph nodes. This is followed by a primary viremia, which disseminates the virus to reticuloendothelial organs such as the liver and spleen. A subsequent secondary viremia results in cutaneous involvement, producing the characteristic centrifugal rash. The lesions appear in successive crops, evolving from macules and papules to vesicles, pustules, and eventually crusts over a period of 3–7 days. (8), (9)
After resolution of the primary infection, VZV establishes latency in sensory ganglia, including the dorsal root and trigeminal ganglia. The viral genome persists within neuronal cell bodies or ganglionic satellite cells. Reactivation can occur in any sensory ganglion, giving rise to herpes zoster. In descending order of frequency, thoracic dermatomes are most commonly affected, followed by cranial, cervical, lumbar, and sacral dermatomes. Although the precise mechanism of reactivation remains unclear, diminished cell-mediated immunity is recognized as a key contributing factor.
Upon reactivation, viral replication occurs within the dorsal root ganglion. Newly formed virions travel via axonal transport to the skin or mucous membranes. This neuronal spread accounts for the prodromal symptoms of pain, dysesthesia, and tenderness, followed by the typical unilateral dermatomal eruption of herpes zoster. Involvement of the nasociliary branch of the trigeminal nerve may produce lesions at the tip of the nose (Hutchinson’s sign), reflecting the shared sensory innervation of the cornea and nasal tip.
Herpes zoster ophthalmicus (HZO) refers to involvement of the ophthalmic division of the fifth cranial nerve by VZV. The ophthalmic division further divides into the frontal, lacrimal, and nasociliary branches. Among these, the frontal nerve is most frequently affected, whereas the lacrimal branch is least commonly involved. The nasociliary nerve supplies the anterior and posterior ethmoidal sinuses, the skin of the eyelids and nasal tip, as well as the conjunctiva, sclera, cornea, iris, and choroid. Hutchinson first described the clinical features of HZO in 1865. Classically, Hutchinson’s sign—defined as vesicular lesions on the tip, side, or root of the nose—is considered a strong predictor of ocular inflammation and corneal sensory loss in patients with HZO.
Brief Summary of CMV infections of the Eye:
Cytomegalovirus (CMV) is a double-stranded DNA virus belonging to the herpesvirus family. It is a large, enveloped virus with a broad cellular tropism. Humans are the only known natural host. Like other herpesviruses, CMV establishes lifelong latency following primary infection. Transmission occurs through close contact with infected body fluids, including saliva, urine, blood, and sexual secretions. (11), (12)
Primary CMV infection usually occurs through contact with infected body fluids. In immunocompetent individuals, it is often asymptomatic or presents as a mild systemic illness. The virus initially replicates in epithelial cells and leukocytes at the site of entry, followed by hematogenous dissemination. CMV has a broad cellular tropism and can infect endothelial cells, epithelial cells, smooth muscle cells, and leukocytes. (11), (12)
CMV replication occurs within the host cell nucleus. After viral entry, the genome is transported to the nucleus, where transcription and DNA replication occur in a regulated sequence (immediate-early, early, and late gene expression). Newly formed nucleocapsids assemble in the nucleus, acquire their envelope during egress, and are released to infect adjacent cells. Viral spread occurs predominantly through direct cell-to-cell transmission. (11), (12)
Following primary infection, CMV establishes latency primarily in monocytes, macrophages, and CD34+ hematopoietic progenitor cells in the bone marrow. During latency, the viral genome persists without active production of infectious virions. The virus remains transcriptionally silent except for limited latency-associated gene expression. Latency is maintained by effective host cell-mediated immunity. Reactivation occurs when cellular immunity is compromised, such as in HIV infection, organ transplantation, or systemic immunosuppression. Under these conditions, latent CMV resumes active replication. Reactivated virus spreads hematogenously and may involve end organs, including the retina. In the eye, viral replication within retinal cells leads to CMV retinitis, characterized by progressive retinal necrosis. (11) Recurrence is common if immune function remains impaired, as antiviral therapy suppresses viral replication but does not eliminate latent virus.
Site of Dormancy:
- HSV-1/2 and VZV: Neurotropic, residing predominantly in neuronal nuclei of trigeminal ganglia.
- CMV: Predominantly in hematopoietic cells (CD34+ progenitors, monocytes, macrophages), forming dendritic/myeloid precursors; possible minor roles in salivary glands or endothelial cells, Lymphoreticular Cells
Some Key Differences betwen HSV,VZV and CMV:
|
HSV, VZV |
CMV |
|
Immunocompetent patients |
Immunocompromised patients |
|
Enters the eye through neurones Typically starts at periphery, a pattern that may corresponds to the termini of ganglion cells of the nerve fibre layers |
Enters the eye through haematogenous route Vasocentric Lesions |
|
Replicates very fast Rapid progression with extensive destruction of retina within a short time |
Replicates very slowly (approx. every 18 hours) Slower progression |

