The ‘Trojan’ Bug
The Trojan Horse is a story from the Trojan War, waged by the Greeks against the city of Troy. After a fruitless siege lasting 10 years, the Greeks constructed a huge wooden horse and hid a few soldiers inside it. As the Greeks appeared to sail away, the Trojans emerged from the city, celebrating their apparent victory, and pulled the horse into the city as a souvenir of triumph. That night, the Greek soldiers emerged from the horse and opened the gates for the rest of the Greek army, which was hiding nearby. The Greeks finally won the battle and destroyed the city of Troy with the help of this deception.
Metaphorically, the term “Trojan Horse” has come to mean any strategy that deceives a target through an outwardly benign appearance. Infection by Toxoplasma gondii follows a similar “Trojan horse” principle. There are three infectious stages of Toxoplasma gondii: the rapidly replicating tachyzoite, the slower-growing bradyzoite (within tissue cysts), and the sporozoite (within oocysts, the eggs of the parasite). Bradyzoites are the dormant or “sleeping” form residing within cysts. Tachyzoites are the active, rapidly multiplying form, primarily responsible for invasion and widespread destruction of ocular tissue.
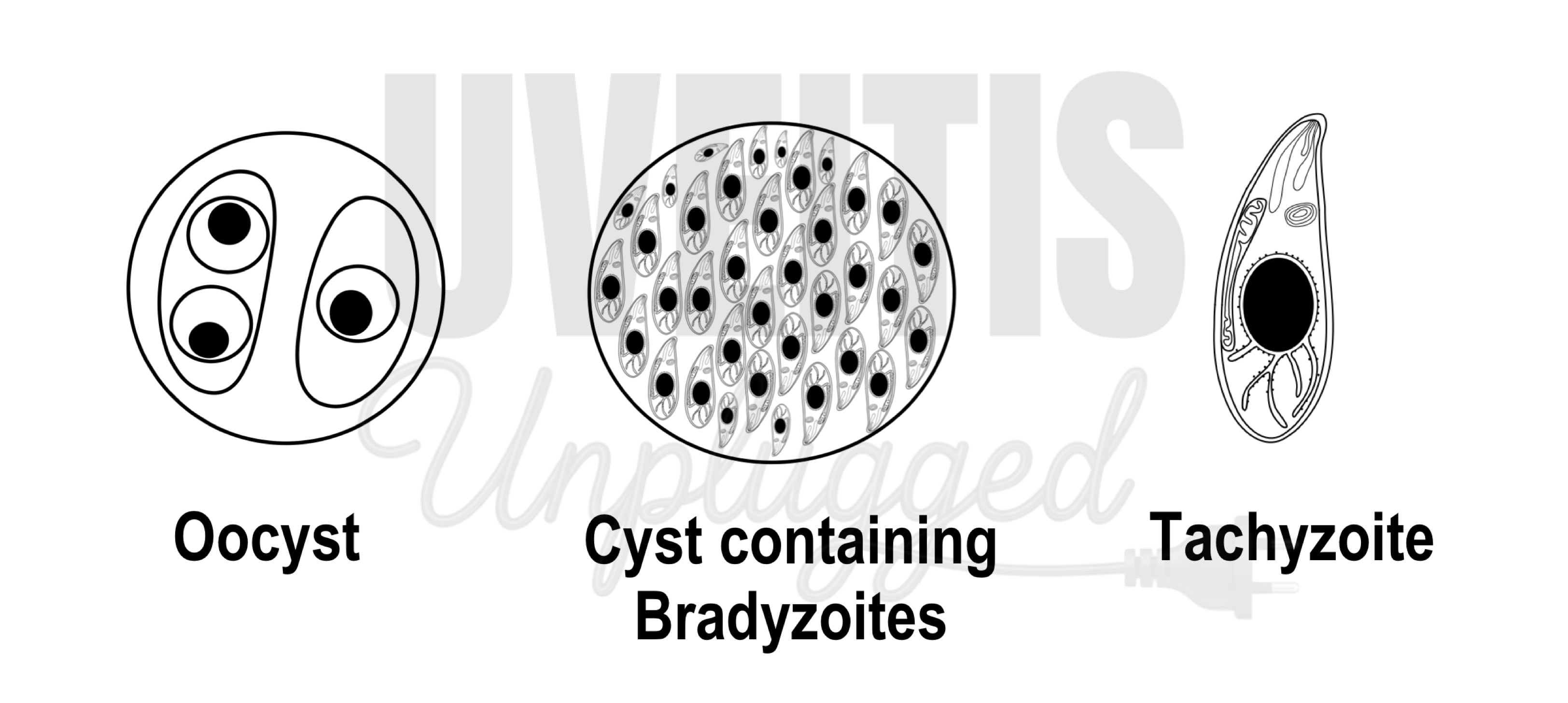
The primary defense against tachyzoites is the interferon-gamma (IFN-γ)–dependent cell-mediated immune response, which eliminates the majority of tachyzoites. (1) Activation of this immune response leads to “cocooning” of the parasite—tachyzoites transform into bradyzoites, which subsequently form cysts. These cysts can remain dormant in the retina for variable periods without causing inflammation or tissue destruction. Each cyst is estimated to contain hundreds to thousands of bradyzoites. Cyst formation can occur in any tissue but is most frequently observed in neural tissues (central nervous system, retina) and muscular tissues (skeletal and cardiac muscle). In the eye, retinal cysts containing hundreds to thousands of bradyzoites remain dormant and are practically resistant to destruction by currently available therapeutic agents. At present, no drugs can eliminate these cysts completely. (2) Many believe that the parasite can modulate the host immune response, allowing it to persist silently for years.

Like the soldiers inside the Trojan horse, bradyzoites within cysts wait for an opportunity to attack. Under favorable conditions, particularly when the immune system is weakened, they emerge from the cysts, transform back into tachyzoites, and invade ocular tissue. Reactivation, especially near old lesions, is therefore common in Toxoplasma gondii infection. Perhaps because of this “Trojan Horse” strategy, this obligate intracellular protozoan parasite is considered one of the most successful parasites worldwide. (3) Toxoplasma gondii can infect almost all warm-blooded vertebrates and has a remarkably diverse host range. (4) It is estimated to infect nearly one-third of the global human population, although prevalence varies widely between countries. The disease burden is significant in many developed regions. In the United States, toxoplasmosis is the second leading cause of death from foodborne illness, and over one million people are infected annually in Europe.
This banana-shaped parasite was discovered independently in 1908 by Nicolle and Manceaux in Tunisia and by Alfonso Splendore in São Paulo, Brazil. Nicolle and Manceaux identified it in a North African rodent, the gundi (Ctenodactylus gundi), while Splendore discovered it in rabbits. The parasite was named Toxoplasma gondii after the curved shape of its infectious stage (from the Greek “toxon,” meaning bow). Cats and other members of the family Felidae are responsible for spreading Toxoplasma gondii into the environment. They excrete oocysts in their feces, which can infect humans. These oocysts contain sporozoites. Humans can become infected by ingesting soil, water, or plant material contaminated with oocysts, as well as by consuming undercooked contaminated meat. Freshwater contamination is an important and often underrecognized route of infection, particularly in populations that consume untreated surface water. (5)
In a report by Hall et al., comparing Toxoplasma gondii antibody prevalence among Jains, vegetarian and non-vegetarian non-Jain Hindus, and other religious groups in a sample of 251 pregnant women in Pune, drinking water was identified as the main vehicle of infection among Jains, who showed significantly higher seroprevalence. (6) In another study from South India, municipal drinking water was implicated in a presumed outbreak of toxoplasmosis presenting as acquired retinitis in 248 patients. (7) Ocular toxoplasmosis remains one of the leading causes of posterior uveitis in retrospective studies from tertiary eye care centers in India. (8),(9) However, not everyone infected develops ocular manifestations. For instance, although approximately 1,075,242 persons are infected with toxoplasmosis annually in the United States, only about 21,000 develop ocular lesions, and approximately 4,800 develop symptomatic ocular disease each year. (10) Host genetic factors are believed to play an important role in determining ocular involvement. (11), (12)
Despite significant advances, we still do not fully understand the precise molecular mechanisms underlying its pathogenesis, nor do we have a drug capable of completely eradicating Toxoplasma gondii or preventing recurrence. Even more than a century after its discovery, this “Trojan bug” continues to menace the sight-saving fraternity.
References:
1. Sturge CR, Yarovinsky F. Complex Immune Cell Interplay in the Gamma Interferon Response during Toxoplasma gondii Infection. Infect Immun. 2014;82(8):3090-3097. doi:10.1128/IAI.01722-14 2. Alday PH, Doggett JS. Drugs in development for toxoplasmosis: advances, challenges, and current status. Drug Des Devel Ther. 2017; 11:273-293. doi:10.2147/DDDT.S60973 3. Sullivan WJ, Jeffers V. Mechanisms of Toxoplasma gondii persistence and latency. FEMS Microbiol Rev. 2012;36(3):717-733. doi:10.1111/j.1574-6976.2011. 00305.x 4. Sibley DL, Charron A, Håkansson S, Mordue D. Invasion and Intracellular Survival by Toxoplasma. Landes Bioscience; 2013. https://www.ncbi.nlm.nih.gov/books/NBK6450/. Accessed November 20, 2019. 5. Bahia-Oliveira LMG, Jones JL, Azevedo-Silva J, Alves CCF, Oréfice F, Addiss DG. Highly endemic, waterborne toxoplasmosis in north Rio de Janeiro state, Brazil. Emerg Infect Dis. 2003;9(1):55-62. doi:10.3201/eid0901.020160 6. Hall SM, Pandit A, Golwilkar A, Williams TS. How do Jains get toxoplasma infection? Lancet Lond Engl. 1999;354(9177):486-487. doi:10.1016/s0140-6736(99)02587-8 7. Balasundaram MB, Andavar R, Palaniswamy M, Venkatapathy N. Outbreak of acquired ocular toxoplasmosis involving 248 patients. Arch Ophthalmol Chic Ill 1960. 2010;128(1):28-32. doi:10.1001/archophthalmol.2009.354 8. Das D, Biswas J, Ganesh SK. Pattern of uveitis in a referral uveitis clinic in India. Indian J Ophthalmol. 1995;43(3):117-121. 9. Rathinam SR, Namperumalsamy P. Global variation and pattern changes in epidemiology of uveitis. Indian J Ophthalmol. 2007;55(3):173-183. 10. Jones JL, Holland GN. Annual Burden of Ocular Toxoplasmosis in the United States. Am J Trop Med Hyg. 2010;82(3):464-465. doi:10.4269/ajtmh.2010.09-0664 11. Peixoto-Rangel AL, Miller EN, Castellucci L, et al. Candidate gene analysis of ocular toxoplasmosis in Brazil: evidence for a role for toll-like receptor 9 (TLR9). Mem Inst Oswaldo Cruz. 2009;104(8):1187-1190. doi:10.1590/s0074-02762009000800019 12. Dutra MS, Béla SR, Peixoto-Rangel AL, et al. Association of a NOD2 gene polymorphism and T-helper 17 cells with presumed ocular toxoplasmosis. J Infect Dis. 2013;207(1):152-163. doi:10.1093/infdis/jis640
